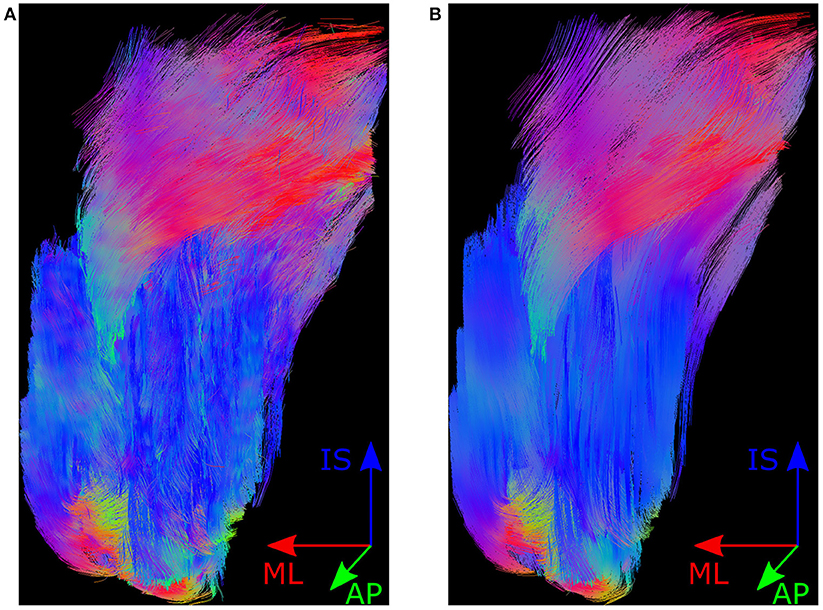
WBSS and TFAS are useful techniques to localize TDP-43 G298S associated alterations over time in this ALS mouse model, as a biological marker.ĭiffusion tensor imaging (DTI) has become an important tool to study the anatomy of the mouse brain in vivo, and its non-invasive nature enables longitudinal studies of transgenic murine disease models. ConclusionĭTI mapping of TDP-43 G298S mice demonstrated progression in motor areas M1/M2. Neuronal loss in layer V of motor cortex was detected in TDP-43 G298S at the later (but not at the earlier) timepoint compared to wt. ResultsĪlterations at the cohort level in TDP-43 G298S mice were observed cross-sectionally and longitudinally in motor areas M1/M2 and in transcallosal fibers but not in the corticospinal tract. Results were supplemented by tractwise fractional anisotropy statistics (TFAS) and histological evaluation of motor cortex for signs of neuronal loss. Whole brain-based spatial statistics (WBSS) of DTI-based parameter maps was used to identify longitudinal alterations of TDP-43 G298S mice compared to wt at the cohort level. 
Ten mice with TDP-43 G298S overexpression under control of the Thy1.2 promoter and 10 wild type ( wt) underwent longitudinal DTI scans at 11.7 T, including one baseline and one follow-up scan with an interval of about 5 months. TBS = Tris, pH 7.In vivo diffusion tensor imaging (DTI) of the mouse brain was used to identify TDP-43 associated alterations in a mouse model for amyotrophic lateral sclerosis (ALS). Careful attention to maintaining equivalent sample volumes is often the most practical approach. Residual formic acid interferes with most protein assays and protein levels may not be proportional to starting materials in any case. Normalizing between samples is problematic. 
Neutralized samples should have a final in assay dilution of at least 10- fold. Indicator should be added to the neutralization buffer to signal when pH 7 is reached.Īssay immediately - DO NOT FREEZE neutralized samples (precipitates of highly enriched insoluble hydrophobic species will form that interfere with assay signal).Ĭare should be taken to have equivalent pH for all samples. If dried completely formic acid brain samples form a plastic-like pellet that is highly resistant to resolubilization.Īliquots of the remaining material must be neutralize (to pH 7) with 5 M NaOH in 1M Tris. The bulk of the formic acid buffer needs to be evaporated off (down to 10-20% of original vol) with a nitrogen stream. Steps to prepare formic acid extracts for Aβ assay Normalization between samples is also problematic. HOWEVER, a difficult multi-step process is required to prepare formic acid fractions for assay (both E1 and whole tissue extracts). For mature β-amyloid deposits in human and old mice brain the highest extraction yields are still observed with this buffer. Hence, E1 fractions with SDS should have a final in assay dilution of at least 3-fold.Įarly protocols used formic acid to extract Aβ from AD brain. Biolegend Aβ kits are resistant to SDS and samples with ≤ 0.2% SDS can be used with minimal loss in sensitivity (< 10%). HOWEVER, SDS is not compatible with many ELISA systems. The ionic detergent SDS gives more complete Aβ extraction from insoluble pellets (> 90% with young and ≈ 50% for older mice). Biolegend's Aβ ELISA's (and most other ELISA's) are compatible with non-ionic detergents. mice > 18 months) with heavy mature β-amyloid deposition. Proportion drops rapidly 20% or less in old animals (i.e. Remove supernatant (S1 or soluble fraction) - usually contains 12 months) up to 80% of total mouse brain Aβ is extracted by Triton X-100.Spin for 20 minutes 350,000xg (or equivalent).EDTA 1 (2mM) can be a useful addition to homogenization buffers (see notes below). Teflon/glass homogenizer with 5-6 passes on ice. Homogenize in TBS with protease inhibitors (Pierce sells excellent solid tablets for use with homogenization buffers) at 5mLs per 1g tissue.Soluble and insoluble tissue fractionation - This protocol generates soluble and insoluble subcellular fractions for analysis of Aβ partitioning in tissues. Preparation of Brain Samples for LEGEND MAX™ Beta Amyloid ELISA
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
